The morning they found it in blood#
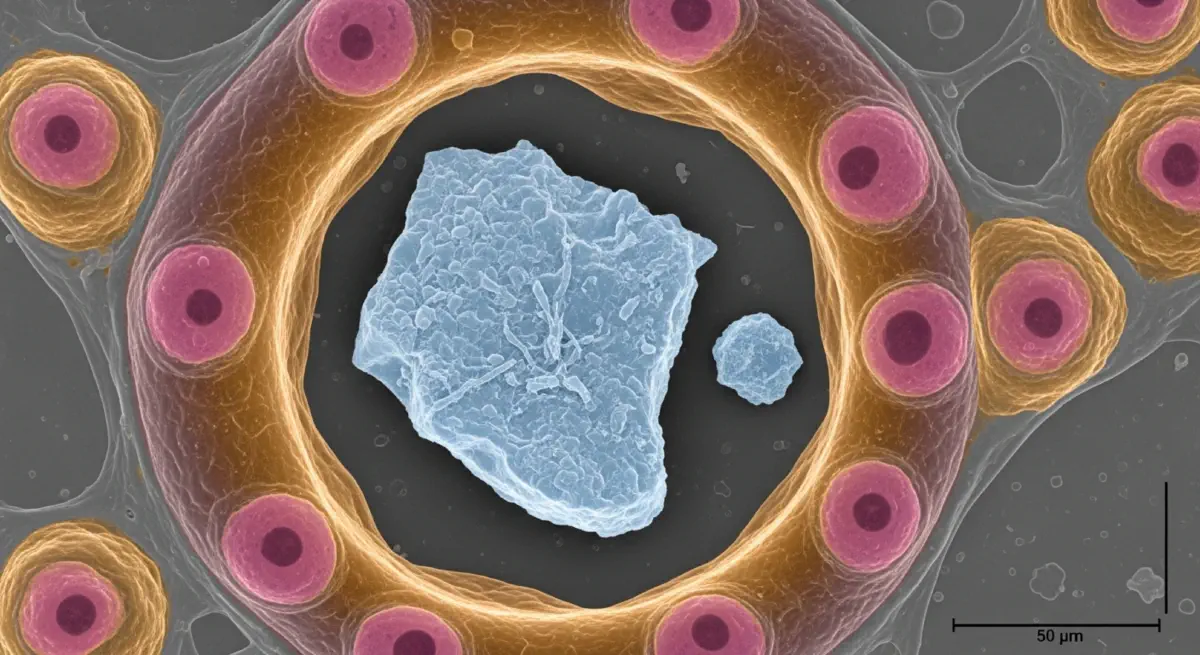
On March 24, 2022, a paper appeared in Environment International that documented, for the first time, the presence of microplastic particles in human blood. The study, led by Heather Leslie at the Vrije Universiteit Amsterdam, analysed blood samples from 22 healthy adult volunteers in the Netherlands using micro-pyrolysis gas chromatography–mass spectrometry — an analytical technique capable of identifying specific polymer types present at concentrations far below visible detection. The result: microplastics were detectable in 17 of the 22 samples (77%). The most common polymer found was PET (from food packaging and textiles); polyethylene and polystyrene were also detected. The mean concentration was approximately 1.6 µg/mL — a mass roughly equivalent to a few hundred plastic particles per millilitre of blood.
The study was immediately cautious in its interpretation. The sample size was small; the analytical method was new; the health consequences of microplastics at detected blood concentrations were unknown. Its significance was not in what it proved about harm — it proved nothing definitive about harm — but in what it documented about exposure: that plastic particles were not merely contaminating environmental matrices (which had been well-established since 2015) but were circulating in the human cardiovascular system. The barrier between what humans put into the world and what was now inside humans had been crossed.
The exposure pathway and the gap in knowledge#
The Plastic Cost Coverage Ratio for the human health externality of plastic is, of its component parts, the most difficult to calculate with precision and the most likely to be systematically understated. The difficulty is not primarily methodological — it reflects genuine scientific uncertainty about the health effects of microplastic and nanoplastic exposure at human tissue-level concentrations. That uncertainty is itself a product of the historical pattern of plastic regulation: plastics were regulated as food contact materials and consumer products based on the direct chemical leachates from intact polymer surfaces, not on the basis of the particle toxicology of their degradation products. By the time analytical chemistry had advanced sufficiently to detect nanoplastics in biological tissue, the exposure had been occurring for decades in an epidemiological vacuum.
The exposure pathway is now documented across multiple routes. Inhalation is the primary route for occupational exposure (textiles workers, plastic manufacturing workers) and a non-trivial route for general population exposure: airborne microplastic concentrations have been measured in urban environments at approximately 0.3–1.5 fibres/m³ and in indoor environments at 1–60 fibres/m³, with synthetic textile fibres the dominant source. Ingestion is the dominant route for non-occupational exposure: microplastics have been documented in bottled and tap water, sea salt, seafood, fresh fruits and vegetables, and beer — essentially every analysed food category. Dermal absorption is plausible for nanoplastic particles but has not been systematically quantified.
Where the particles travel after absorption is now partially documented. The 2022 Leslie bloodstream study established systemic circulation. A 2021 Italian paper by Pironti and colleagues documented microplastics in human placental tissue. A 2023 study, published in Environmental Health Perspectives, documented nanoplastic particles (below 200 nanometres) in brain tissue in post-mortem samples. The anatomical progression — environment to gut to bloodstream to placenta to brain — represents a toxicological scope that regulatory science has not yet fully assessed.
What particle toxicology shows#
The toxicology of microplastic and nanoplastic particles is studied through in vitro cell culture experiments, in vivo animal models, and ex vivo tissue studies. The findings, while not generating clean dose-response curves at human-relevant exposure levels for chronic outcomes, consistently show biologically active effects at concentrations comparable to or below those now detectable in human tissue:
Oxidative stress — the generation of reactive oxygen species that damage cell membranes, DNA, and proteins — is the most consistently documented cellular effect of microplastic particle exposure across a wide range of polymer types. Studies reviewing multiple cell types (lung epithelial, intestinal epithelial, human blood cells, hepatocytes) find significant oxidative stress markers at particle concentrations of approximately 5–100 µg/mL — a range not categorically different from the concentrations detected in human blood by the Leslie study.
Inflammatory response — sustained low-grade pro-inflammatory cytokine production — is the second most consistently documented effect. Chronic low-grade inflammation is the mechanistic pathway through which PM2.5 air pollution generates cardiovascular mortality (the pathway that makes coal-power mortality estimation reliable); if microplastics generate comparable inflammatory signals in cardiovascular tissues, the mortality consequence would follow similar epidemiological dynamics — diffuse, statistical, and not attributable to specific exposures in individual cases.
The 2023 study by Rubio and colleagues in the Journal of Hazardous Materials found DNA damage (genotoxicity) in human haematopoietic cell lines at nanoplastic concentrations. DNA-damaging agents in circulating blood cells represent an elevated cancer risk pathway. The epidemiological evidence connecting measurable polystyrene nanoparticle exposure to cancer incidence in human populations does not yet exist — the exposure only became analytically detectable at tissue levels in the 2020s — but the biological chain of events from exposure to genotoxic damage is mechanistically plausible and experimentally supported.
The additive chemistry exposure#
The particles themselves are not the only relevant exposure pathway. Plastic additives — the phthalates, bisphenol compounds, flame retardants, UV stabilisers, and other chemicals incorporated into polymer matrices — are not chemically bonded to the polymer backbone and leach continuously from intact plastic surfaces, not just from degradation fragments. The phthalate and BPA epidemiology is the most extensively studied component of plastic chemical exposure.
Phthalate urinary metabolite concentrations are detectable in essentially all populations studied in high-income countries. The US NHANES (National Health and Nutrition Examination Survey) data show measurable phthalate metabolites in over 95% of adult Americans. A 2018 US study by Zota and colleagues found that approximately 80% of Americans had detectable serum BPA; the decline in BPA concentrations following regulatory action has been partially offset by increases in structurally similar bisphenol substitutes that were introduced without prior toxicological review of the specific applications.
The body burden of plastic-associated chemicals includes compounds across reproductive toxicity, developmental toxicity, and endocrine disruption categories. The cumulative exposure to multiple chemicals with similar mechanisms of action — multiple phthalates acting simultaneously on the same receptor systems — is not additive in a simple linear sense; it can be additive, synergistic, or antagonistic depending on the specific compounds and target pathways. Regulatory toxicology has generally assessed compounds individually, in single-substance risk assessments, rather than in the mixture conditions that characterise actual human exposure.
The precautionary arithmetic#
The precautionary arithmetic of the microplastic health externality does not require proof of specific mortality effects at current exposure levels to generate a policy-relevant finding. It requires only the following components, all empirically supported: microplastics are present in human blood and brain tissue; particle exposure generates oxidative stress, inflammation, and DNA damage in human cell lines at concentrations comparable to detected tissue levels; the epidemiological infrastructure to detect chronic health outcomes from this exposure has not existed long enough to generate population-level disease data; and the PCCR for chemical and particle health externalities in plastic production is approximately zero.
The policy implication of the precautionary arithmetic is the same whether the eventual epidemiology is mild or severe. A chemical or material that is present systemically in essentially all members of the global population, that generates biologically active effects at detected concentrations in cellular models, and whose long-term health consequences are unknown, is a material whose production warrants the precautionary principle — the regulatory standard that requires a reasonable basis for safety before deployment at scale, rather than a waiting for proof of harm after the fact.
Plastic has been deployed at scale — 400 million tonnes per year — without ever being assessed for the safety of the particle-exposure consequences of its end-of-life degradation. The PCCR captures the financial expression of this regulatory gap: zero liability for health consequences that were never assessed before the product was released into the environment at a rate that has now contaminated the bloodstream of every measurable human population on Earth.
The next and final post in this series examines the political economy of changing the Plastic Cost Coverage Ratio — the EU Plastics Strategy, EPR regimes, and most consequentially, the Global Plastics Treaty negotiation that began in 2022 and whose outcome will determine whether plastic production is governed by a mandatory international framework or whether the invisible invoice continues to go unpaid.