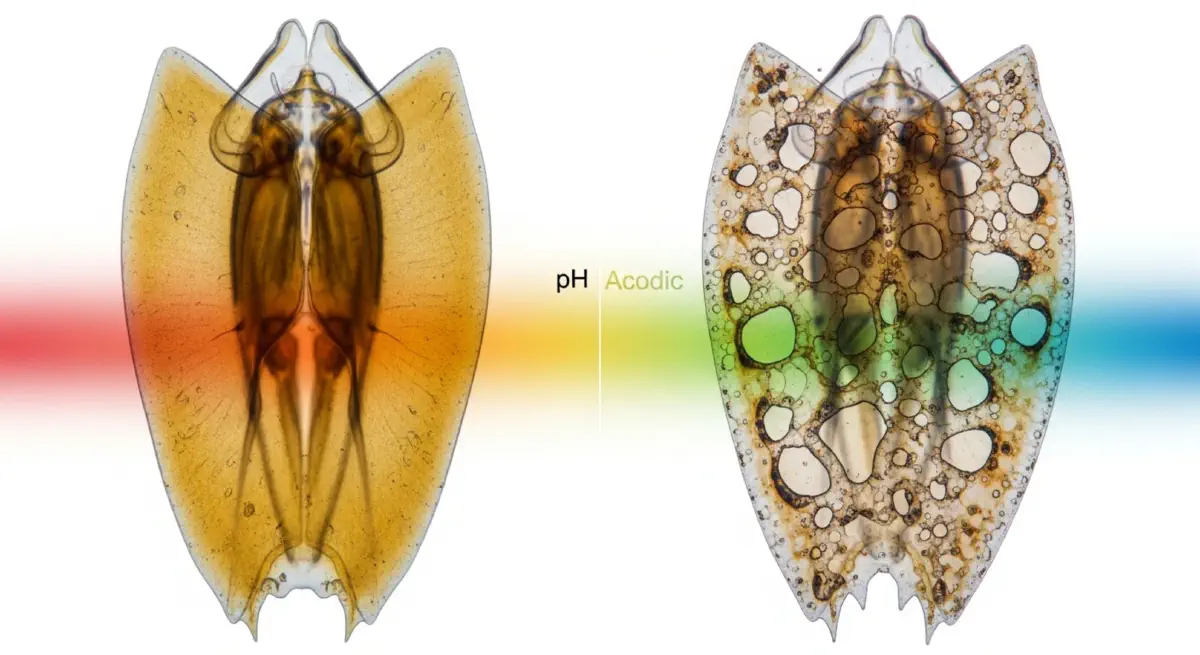
What a tenth of a pH unit means#
In March 2005, Ken Caldeira and Michael Wickett published a short paper in Nature that contained a sentence climate scientists had understood for decades but that had never appeared in a major journal with such directness: "the ocean has absorbed a substantial fraction of man-made carbon dioxide emissions, and the resultant changes in ocean chemistry are likely to decrease the calcification rates of key marine organisms." They were not describing a possible future effect — they were describing a process already well under way. The ocean had absorbed approximately 118 billion tonnes of anthropogenic carbon between 1800 and 2005. The chemistry of that absorption involves carbonic acid formation. The ocean was already more acidic than it had been in at least 400,000 years.
The numbers look small on first reading. Pre-industrial ocean surface pH averaged approximately 8.2. By 2024, it had declined to approximately 8.08. The difference is 0.12 pH units — a figure that sounds negligible until the logarithmic nature of pH is applied. pH is the negative logarithm of hydrogen ion concentration. A decline of 0.1 pH units represents a 26% increase in hydrogen ion concentration. In pH terms, the ocean is 30% more acidic than it was before industrialisation. Under the RCP 6.0 scenario — a moderate emissions trajectory — ocean surface pH is projected to reach approximately 7.95 by 2100; under RCP 8.5, approximately 7.8. The last time the ocean was at pH 7.8 was approximately 50 million years ago, when atmospheric CO₂ concentrations were 1,000–2,000 parts per million.
An unbooked liability accumulating on the global balance sheet#
The ocean's carbon sequestration service — absorbing approximately 25% of annual anthropogenic CO₂ emissions — is one of the most valuable ecosystem services the planet provides. It has also, over the course of its operation, generated a liability that now threatens the ecosystem services it is part of. The chemistry is fixed: dissolved CO₂ reacts with seawater to form carbonic acid (H₂CO₃), which dissociates to hydrogen ions and bicarbonate, reducing the availability of carbonate ions that marine organisms require to build calcium carbonate shells and skeletons. The ocean cannot continue absorbing carbon without becoming more acidic, and more acidic conditions progressively compromise the organisms that make the marine food web function.
This is the structural tension at the core of the Marine Extraction Ratio as it applies to ocean acidification: the ocean's most valuable service — carbon absorption — is operating through a mechanism that degrades other ocean services. The liability is not hypothetical, not future, and not small. It is currently accumulating at approximately 2.5 billion tonnes of carbon per year. None of it is on any government's balance sheet.
The chemistry of shells#
Calcium carbonate exists in two mineral forms that marine organisms use to build structures: calcite (used by most coccolithophores — the microscopic algae that form the base of open-ocean food webs — and many foraminifera) and aragonite (used by corals, pteropods, and some oysters). Aragonite is approximately 50% more soluble than calcite, meaning it dissolves at higher pH levels. The "aragonite saturation state" — a ratio describing whether the ocean is supersaturated (supporting shell formation) or undersaturated (causing dissolution) — has declined globally by approximately 16% since pre-industrial times.
In the Southern Ocean and parts of the Arctic Ocean, aragonite undersaturation occurs seasonally at depths as shallow as 200 metres. The pteropods — small, free-swimming molluscs that are a critical prey species for salmon, herring, cod, and baleen whales in polar and sub-polar food webs — have been documented with shell dissolution in surface waters off the US Pacific coast since 2008. A 2012 study by Nina Bednarsek and colleagues found that approximately 53% of pteropods sampled in surface waters of the California Current system showed severe shell dissolution — a condition associated with abnormal shell formation and reduced survival rates. Pteropods are not a marginal species. In some parts of the North Pacific and Southern Ocean, they represent 20–90% of the prey volume for species that support commercial fisheries worth tens of billions of dollars per year.
Oyster hatcheries in the US Pacific Northwest began experiencing mass mortality events in 2007. The Whiskey Creek shellfish hatchery in Oregon and Taylor Shellfish in Washington both traced the mortality of larval oyster sets to corrosive upwelling water that was, by pH measurement, undersaturated relative to the aragonite requirements of juveniles. The Pacific oyster industry on the US West Coast generates approximately $270 million per year; it nearly collapsed between 2007 and 2012 before hatcheries developed mitigation strategies including water chemistry monitoring and CO₂ scrubbing of hatchery intake water. The solution was possible at the hatchery scale. It is not possible at the ocean scale.
What acidification does to reefs#
Coral reef ecosystems have been studied longer and with more precision than almost any other marine environment, partly because their economic importance is obvious (coastal protection, tourism, fishery nursery grounds) and partly because their biological response to thermal and chemical stress is visible, rapid, and catastrophic. Global coral reef ecosystem services are estimated at approximately $375 billion per year (Hoegh-Guldberg et al., 2007), distributed across approximately 500 million people who depend on reef fisheries for protein and approximately 150,000 km of coastline where reefs provide measurable storm surge attenuation.
The relationship between ocean pH and coral calcification is quantified by laboratory experiments and field studies with unusual precision. A 2009 meta-analysis by Joan Kleypas and colleagues examined 327 data points from 44 studies and found a robust negative relationship between aragonite saturation and coral calcification rates. Below an aragonite saturation state of approximately 2.2 — projected globally for most tropical reefs under RCP 4.5 by mid-century — coral calcification rates decline to levels at which the structural integrity of reef systems is compromised. Bioerosion exceeds accretion; the reef begins to dissolve faster than coral growth can rebuild it.
The Great Barrier Reef's coral calcification rates, as measured by annual skeletal density bands in coral cores, declined by approximately 14% between 1990 and 2005 (De'ath et al., 2009) — the largest and most geographically extensive decline in calcification on record. The decline correlates with both rising sea surface temperatures and declining aragonite saturation. Temperature and acidification operate synergistically: a coral bleached by thermal stress takes significantly longer to recover if recovering into more acidic water, because calcification rates are constrained by chemistry during the recovery phase.
The marine food web and its pH dependencies#
Acidification is not only a surface-water phenomenon. As atmospheric CO₂ continues to rise, the depth of the aragonite saturation horizon — the depth above which carbonate minerals remain stable and below which they dissolve — rises toward the surface. Between 1990 and 2005, this horizon shoaled by approximately 75–150 metres in the North Pacific Ocean. Organisms whose life cycles span a range of depths are increasingly exposed to undersaturated water during developmental stages that were previously protected by depth.
The biological consequences radiate outward from the organisms directly affected by acidification to every species that depends on them. Pteropod dissolution affects salmon and cod. Coccolithophore disruption — these organisms produce calcite plates that serve as ballast, driving carbon to the deep ocean in marine snow — potentially affects the efficiency of the biological carbon pump, the process by which photosynthetically fixed carbon is exported from surface waters to depth. If the biological pump is disrupted by acidification, the ocean's carbon sequestration efficiency declines — a feedback loop that would accelerate acidification itself.
The total economic value of the marine services threatened by acidification under moderate emissions scenarios exceeds $10 trillion per year by mid-century in estimates that attempt to capture fishery disruption, reef service loss, and coastal protection degradation. Against this figure, the MER for the ocean's carbon absorption service — the implicit "cost" being charged to the ocean for the climate service it provides — is not a ratio between two market prices. It is a ratio between a service provided for free and liabilities accumulating for no one.
The account no one agreed to open#
Ocean acidification is the only major environmental externality of the global economy that does not appear in any binding international agreement, any national carbon price mechanism, or any insurance or financial liability framework. The mechanism is well understood. The relationship between atmospheric CO₂ concentration and ocean pH is directly calculable, reproducible in laboratory conditions, and confirmed by ocean monitoring data from hundreds of sampling stations worldwide. The trajectory is not uncertain — it is a function of emissions, which are themselves a matter of human decision. The damages are real, quantified at scales of hundreds of billions to trillions of dollars per year, and accumulating.
What is absent is any accounting system that records this accumulation as a liability with an identified debtor. Carbon dioxide emissions generate ocean acidification in direct proportion to their volume. Every tonne of CO₂ emitted affects ocean chemistry. The relationship between the emitter and the environmental liability is physically exact — more exact than most pollution pathways in existing regulatory frameworks. The liability is simply not recorded, not because the causal chain is obscure, but because no governance framework has agreed to audit it.
The next post moves from chemistry to geology — from what the ocean is becoming to what lies beneath it. Deep-sea mining represents the next major expansion of marine resource extraction, and its Marine Extraction Ratio, incorporating the ecosystem service value of the seafloor environments that will be destroyed, has never been calculated for any commercial mining proposal. The floor below the ocean floor is about to be opened for business.